DRUP Publication in Clinical Cancer Research
Immune checkpoint blockade (ICB) has shown promise in tumors with high tumor mutational burden (TMB), but the absence of formal EMA approval highlights a lack of strong supporting evidence.
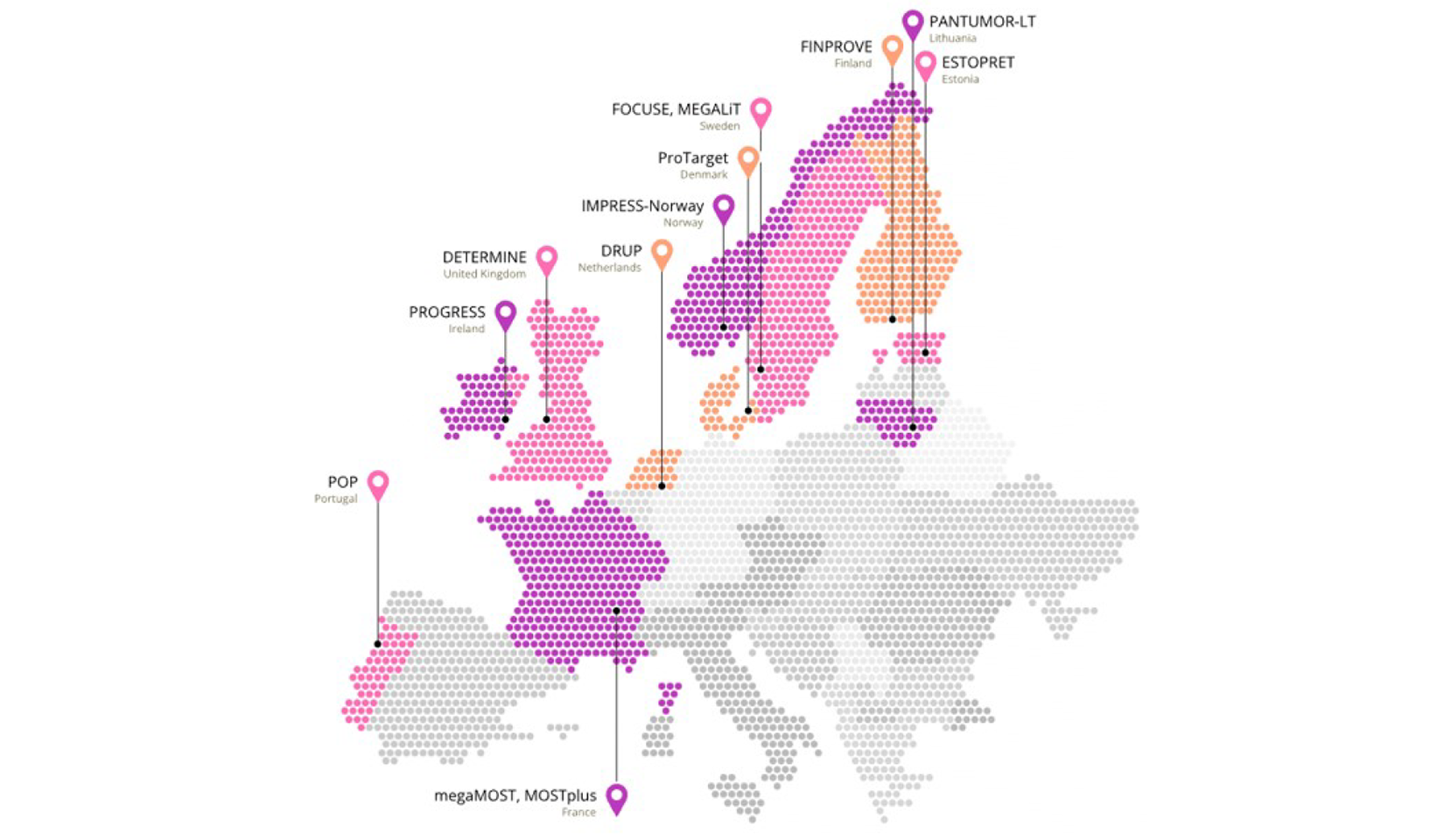
Within the Drug Rediscovery Protocol, we tested the ICB combinations atezolizumab plus bevacizumab and nivolumab plus ipilimumab in patients with TMB-high tumors and found that durable benefit occurred mainly in those with TMB above 20 mutations per megabase.
Early treatment initiation further improved outcomes, while biomarker analysis linked adaptive immune infiltration to longer progression-free survival. These results point to refined TMB cut-offs and immune biomarkers as tools to better guide patient selection for ICB in clinical practice.