Jacqueline Jacobs
Telomere damage and cancer
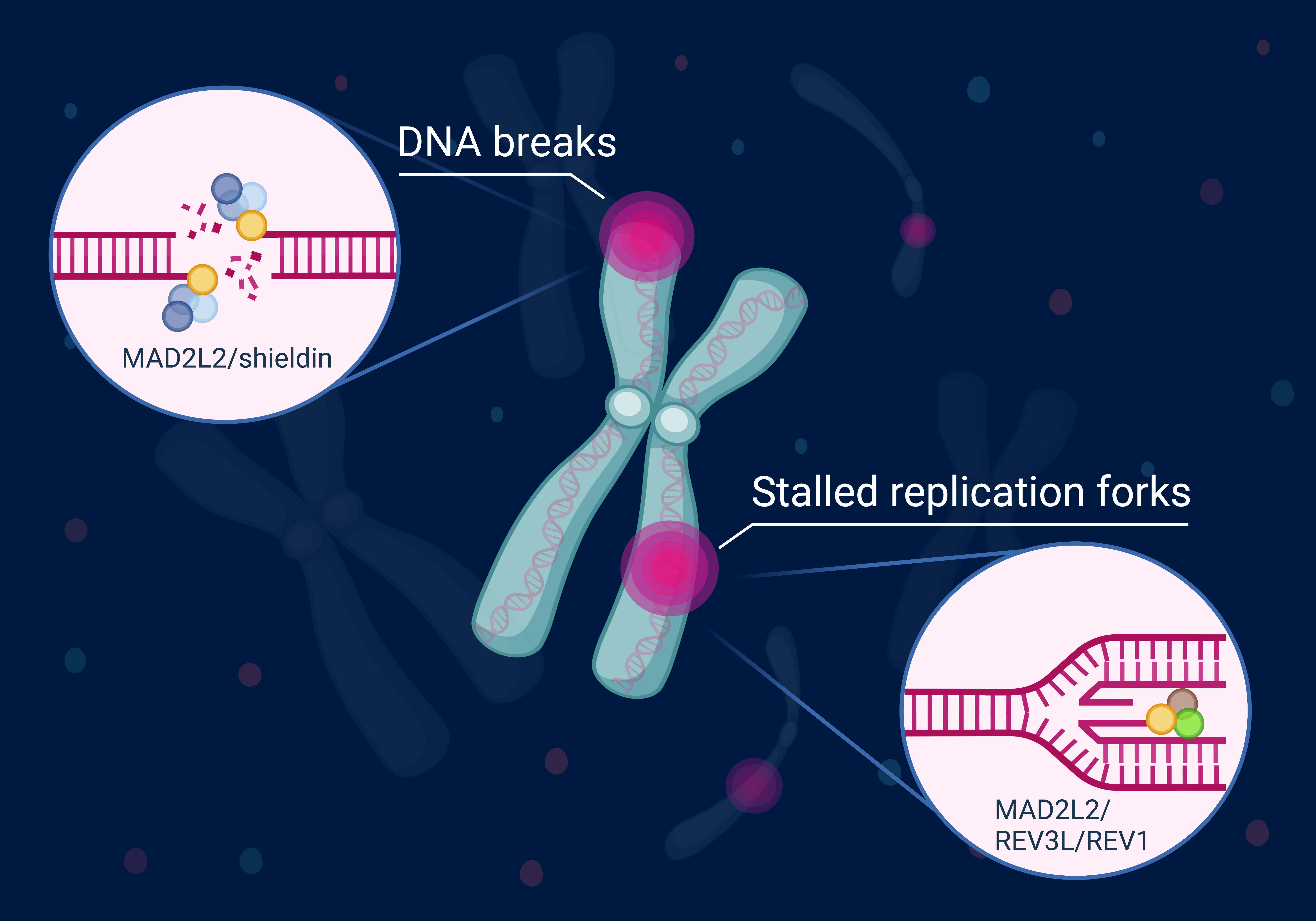
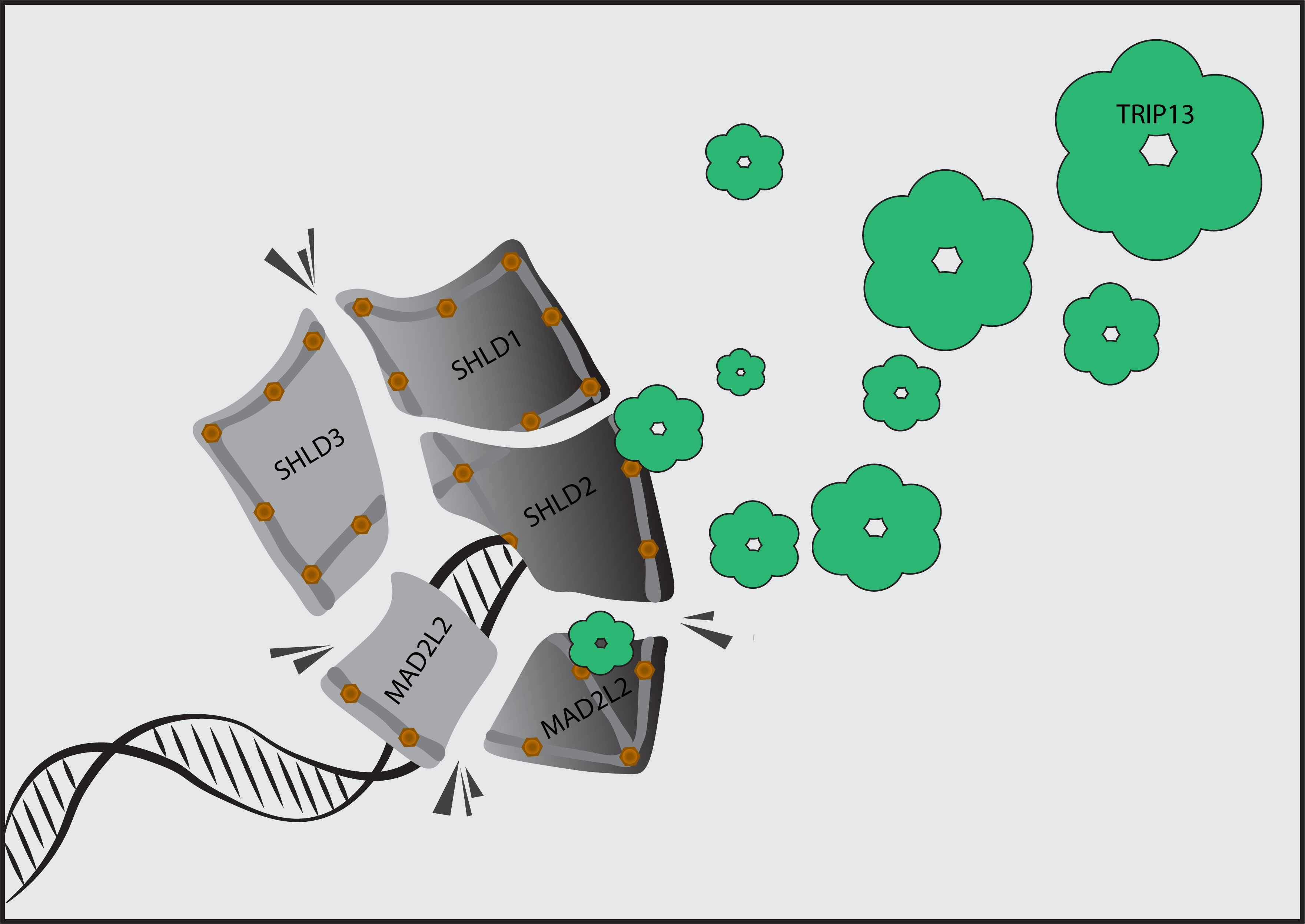
Breaks in our genome need to be efficiently detected and faithfully repaired. At the same time, the natural ends of our chromosomes (i.e. telomeres) should not be seen and processed as broken DNA. Tight control of DNA damage detection and repair is critical for maintaining genome integrity and preventing or treating cancer.
We study the molecular mechanisms underlying this control at telomeres and DNA breaks to understand how genome integrity is maintained, how genomic instability and pathology arise when these mechanisms fail and how these mechanisms affect DNA damaging cancer therapies.