Daniel Peeper
Functional oncogenomics for tumor & immunotherapy
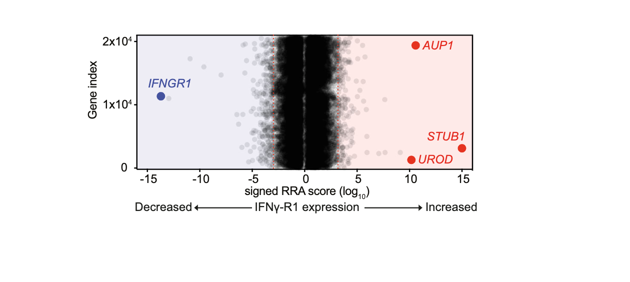
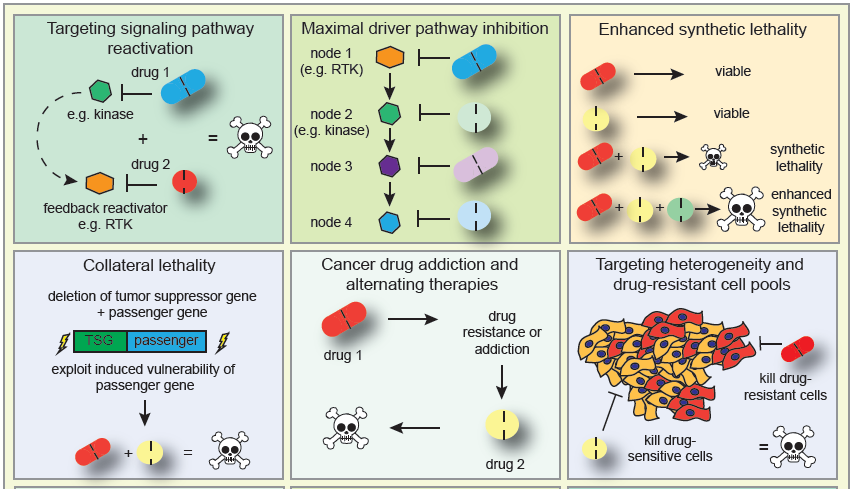
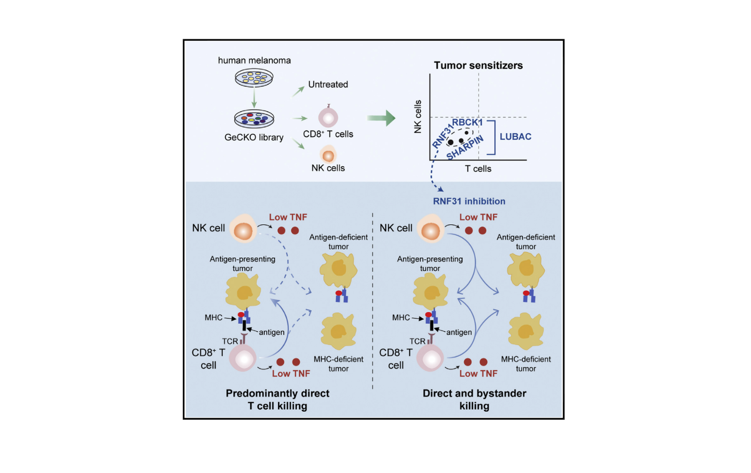
Tumor heterogeneity, immune dysfunction and therapy resistance are among the most substantial challenges that limit durable benefit of cancer therapies. Using powerful function-based genomics, we screen for novel therapeutic targets to tackle those clinical problems.
We develop rational combinatorial cancer treatments, which target both cancer and immune cells, thereby simultaneously eliminating the patient’s tumor and harnessing the immune system. This has already culminated in new concepts that we are translating to the benefit of the patient.
On this page we’re sharing some of the highlights of our lab, news items, our research, team members and, if you scroll to the bottom, our vacancies.