“Our starting point was the high activity we observed with combination immunotherapy in locally advanced bladder cancer,” says medical oncologist Michiel van der Heijden. “In earlier studies, about half of these high-risk patients had no residual tumor after cystectomy. This suggests that powerful induction therapy could create opportunities to treat more patients effectively while preserving the bladder.”
Rather than focusing on a single drug regimen, the team focused on a broader concept: organ preservation enabled by strong induction therapy, followed by consolidation treatment. This concept has been tested in the phase II INDIBLADE trial, which showed that induction combination immunotherapy followed by chemoradiotherapy provides high bladder-intact event-free survival at two years, supporting the feasibility of bladder-sparing treatment in a broader group of patients.
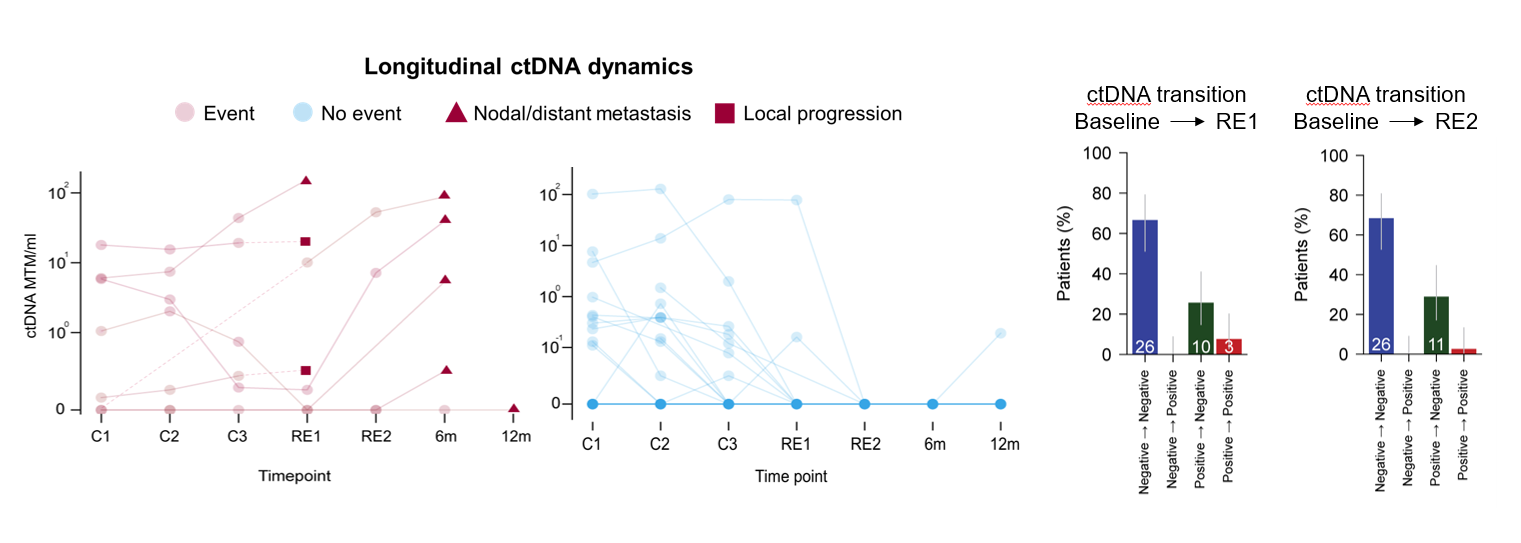
The study is the result of a stepwise, systematic research program: first establishing safety, then optimizing schedules, improving response assessment with advanced imaging and circulating tumor DNA (ctDNA), and finally integrating these elements into a new treatment strategy. The results were presented last weekend at ASCO-GU 2026 and published simultaneously in Nature Medicine.
“The key strength of the study is the concept,” Van der Heijden explains. “The study is not about one specific induction regimen. The exact therapies will continue to evolve and we already have even more promising approaches for the next step. Our goal is to move towards more personalized strategies, and potentially to be able to identify patients who may need less local treatment of the bladder, or perhaps even none at all.”