Our Research
Treatment response
We aim to understand treatment response in model systems and patients. We focus on three themes. In Theme 1, ‘Cell decisions’ we use single cell perturbation and longitudinal profiling to infer the regulatory machinery that controls cellular state transitions. Theme 2, ‘Cell maps’, maps the regulatory circuitry in cells through network inference on large scale screens and experimental validation. Finally, in Theme 3, ‘Treatment design’, we aim to design optimal treatment (combinations) by understanding single and combination treatment response based on pre- and on-treatment measurements.
Theme 1: Cell Decisions
Example project: Single cell perturbation and profiling. We are developing an approach to perform simultaneous, pooled, bar-coded perturbation and molecular profiling in single cells. We will perform proteomic profiling with a panel of 33 DNA-tagged anti-bodies (ABs). Employing combinatorial indexing and the tagged ABs, we will run time-course and perturbation experiments on various cancer cell lines to generate proteomic profiles of single cells under various perturbations. We will employ the algorithms we specifically developed for single cell data, such as single cell MRA, as well as machine learning (deep learning) approaches to reconstruct the cellular decision making machinery.
Theme 2: Cell maps
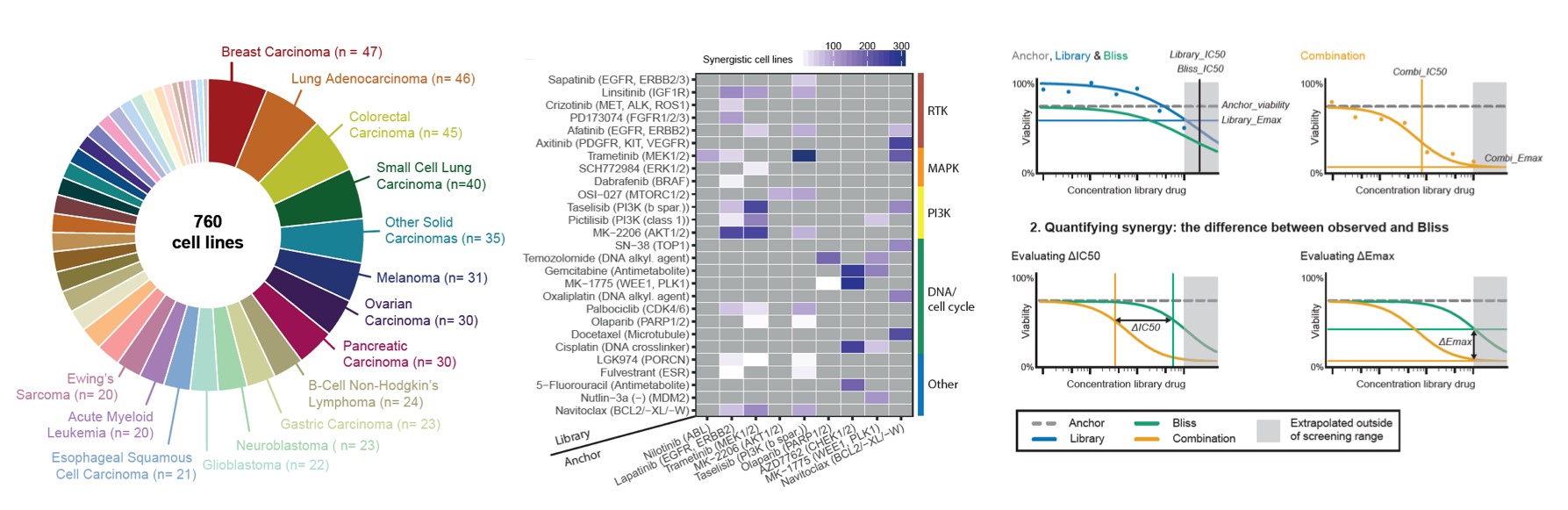
Example project: CellCircuits: A genomics compendium to identify therapeutic targets.
Cells are equipped with a complex decision-making machinery that largely remains poorly understood. Although this machinery is orchestrated by the activity of human genes,
no wiring diagram exists depicting their function. This project will link the function of all
human genes to a compendium of cellular processes. In addition to our computational know-how, this project will draw upon the large collection of haploid screens generated by Scenic Biotech BV (Phenosuarus) and the screening and cell and molecular biology expertise
of the Brummelkamp laboratory. With the screening technology to generate this genomics compendium proven to be highly effective, the opportunity exists now to cover diverse cellular functions and to use computational methods to deduce the underlying genetic control mechanisms. This will suggest new entry points to manipulate cellular processes and therapeutic strategies to rebalance disease.
Theme 3: Treatment design
Example project 1: PRECISE+ predicts drug response in patients by transfer of predictors from pre-clinical models. Pre-clinical models have been the workhorse of cancer research for decades. Albeit powerful, these models do not fully recapitulate the complexity of human tumors. Consequently, translating biomarkers of drug response from pre-clinical models to human tumors has been particularly challenging. To explicitly take these differences into account and enable the efficient exploitation of the vast pre-clinical drug response resources, we developed PRECISE+, a novel transfer learning framework for drug response prediction. On PDXs models, and on human patients from TCGA and the HMF, PRECISE+ outperforms benchmark approaches in predicting response to treatment. PRECISE+ also allows the identification of novel putative biomarkers of resistance to Paclitaxel and Gemcitabine.
Example project 2: Dissecting the mechanisms of Multiple Low Dose (MLD) treatment. The Bernards group demonstrated that partial inhibition of each of the EGFR-RAF-MEK-ERK kinases using a very low doses of inhibitors in EGFR mutant lung cancer, resulted in complete cell killing in long term assays. Escape from this regimen could not be induced by prolonged in vitro culture nor through genome-scale genetic screens. In mouse models, MLD also showed durable tumour control without toxicity. We will investigate the role of pathway architecture, expression heterogeneity of pathway proteins and population heterogeneity in MLD response. We will employ the single cell profiling technology developed in Theme 1, the FRET-pERK sensor (Snippert) and single cell pathway reconstruction based on MRA, to test these hypotheses.